Thermal management of electronics can be simply described as using cool fluids to remove heat from hot devices. But what about these fluids? How do you know which fluid to use for your system? What properties matter for heat transfer and where do you find them? This post is a one-stop-shop for heat transfer fluid related questions.
To begin, let’s do a quick survey of the most common coolants and what their properties are (as a quick aside, I find myself spending too much time searching tables in textbooks and scouring Google to locate fluid properties; now I know the one place I need to look):
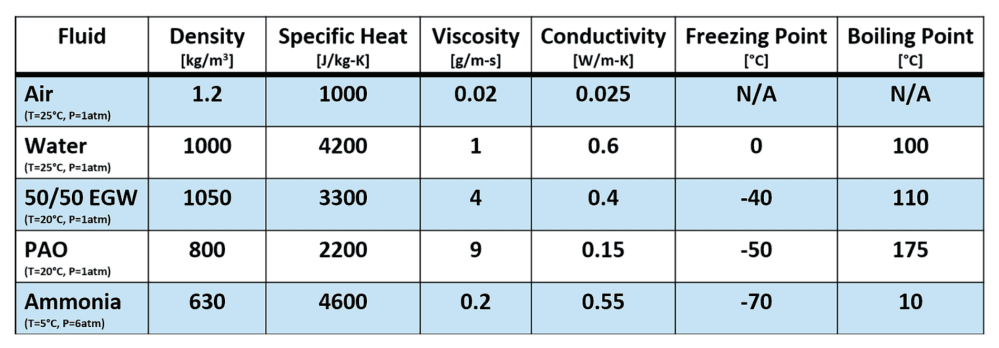
Table 1: Thermofluidic properties for common heat transfer fluids [1,2,3,4]. Note that these fluid properties may vary significantly as a function of temperature and pressure.
The density, specific heat, thermal conductivity, viscosity, boiling point, and freezing point are the key quantities to keep track of when designing a thermal management solution. The phase change points are key in finding the right balance of ambient, device, and coolant temperature. Meanwhile, the thermophysical properties help quantify the heat transfer effectiveness and pumping requirements.
But how do we interpret these different properties for each fluid? Let’s run through a quick breakdown of each fluid and where it is most commonly used.
What Is a Heat Transfer Fluid?
A heat transfer fluid (HTF) is a liquid or gas used to move heat away from a device, component, or system. These fluids circulate through cooling or heating systems, absorbing thermal energy from hot surfaces and carrying it away to maintain safe operating temperatures.
In electronics and data center environments, heat transfer fluids are used in liquid cooling systems to efficiently remove heat from processors, GPUs, and other high-power components.
Common heat transfer fluids include: (link these to headings)
Each fluid offers different thermal properties, operating temperature ranges, and implementation considerations.
Common Heat Transfer Fluids and Their Properties
Air
Air is the most common heat transfer fluid; if your system resides in the atmosphere, air is almost guaranteed to find its way into your heat pathway at some point.
Specifically, air is most effectively used for low power density electronic devices and as an ultimate heat sink. This is because the low fluid density and thermal conductivity values correspond to low heat transfer coefficients, which therefore require a large area for any noteworthy cooling to occur.
As a result, air is frequently used with finned heat sinks, heat pipes, and PCB spreading techniques to augment the cooling area. Air is often used as a coolant in low density data centers, aircraft, automotive radiators, and cell towers.
In short, the versatility of air with its dielectric nature and ubiquity make it a common, low stress choice for low power density systems.
Water
Water is also quite common, and for good reason. It has favorable specific heat, thermal conductivity, and viscosity values, making it an extremely effective cooling fluid. With its enhanced properties, water systems typically achieve 10X the cooling performance of equivalent air systems.
Water is used in high power data centers, heat-dense power electronics applications, and high power and frequency communication systems. The most critical disadvantages of water are its electrical conductivity and corrosive nature, creating challenges in implementation and cooling module design.
But, even if a few additives are required to get it to behave the way you want, water is the #1 choice for those with high power density devices in controlled environments.
50/50 Ethylene-Glycol + Water (EGW) Mix
The other disadvantage of water is that it freezes at a relatively high temperature. For applications that are not in the comfort of the lab or a temperate climate, freezing becomes a major implementation concern.
This is where a 50/50 mix of water and glycol comes in.
You sacrifice some thermal performance, but the freezing point is lowered to make a fluid that is robust to common ambient temperature changes. Water glycol mixes are commonly used in aircraft, automotive applications, and even some computing applications.
Overall, the 50/50 water glycol mix is a happy medium between high performance and temperature survivability.
PAO
Polyalphaolefin (PAO) is more niche, but its properties are reflective of a larger class of mineral oils that are fairly commonly used. The thermal properties of PAO are a significant downgrade from water or even the glycol mix, making it challenging to directly compete with other liquid coolants.
The major advantages of PAO are:
- it is a dielectric liquid
- it has a tremendous range of operating temperatures without freezing or boiling.
Where PAO finds a home is in certain military electronics systems and direct contact cooling of large-scale electronics (e.g. transformers) whose power starts to push the limits of air. This is to say, PAO should not be used unless you really need it – whether due to a difficult geometry or harsh environmental conditions.
Ammonia
Ammonia systems also may not be as commonplace but fill an important role in space systems. Because of ammonia’s terrific freeze protection, it can be used in environments that reach extremely low temperatures.
Additionally, with overall strong thermal properties, it brings very competitive heat transfer rates. Ammonia is not often used in terrestrial applications due to its health and safety concerns, and the pressure needs to be carefully controlled to operate in the proper regime. Ammonia is also used as a two-phase fluid, which is a discussion for another time.
Overall, ammonia is currently limited to space applications (such as on the ISS) and other niche use cases, but is a very serviceable option when implemented properly.
There are other fluid options out there, especially in multiphase cooling applications. But this discussion covers the baseline considerations of heat transfer fluids and where they are most effectively used. When planning out your next thermal management system, begin by scoping out the requirements and expected performance of the fluid you ultimately choose.
Discover how liquid cooling technologies can prepare your data center for high-density computing in our data center, high-performance computing, and colocation case studies.
References
[1] Mohapatra, Satish C. “An Overview of Liquid Coolants for Electronics Cooling.” Electronics Cooling, 2 July 2019, http://www.electronics-cooling.com/2006/05/an-overview-of-liquid-coolants-for-electronics-cooling/.
[2] “NIST Chemistry WebBook, SRD 69.” Thermophysical Properties of Fluid Systems, webbook.nist.gov/chemistry/fluid/.
[3] “Dry Air Properties.” Engineering ToolBox, http://www.engineeringtoolbox.com/dry-air-properties-d_973.html.
[4] “Ethylene Glycol Heat-Transfer Fluid.” Engineering ToolBox, http://www.engineeringtoolbox.com/ethylene-glycol-d_146.html.